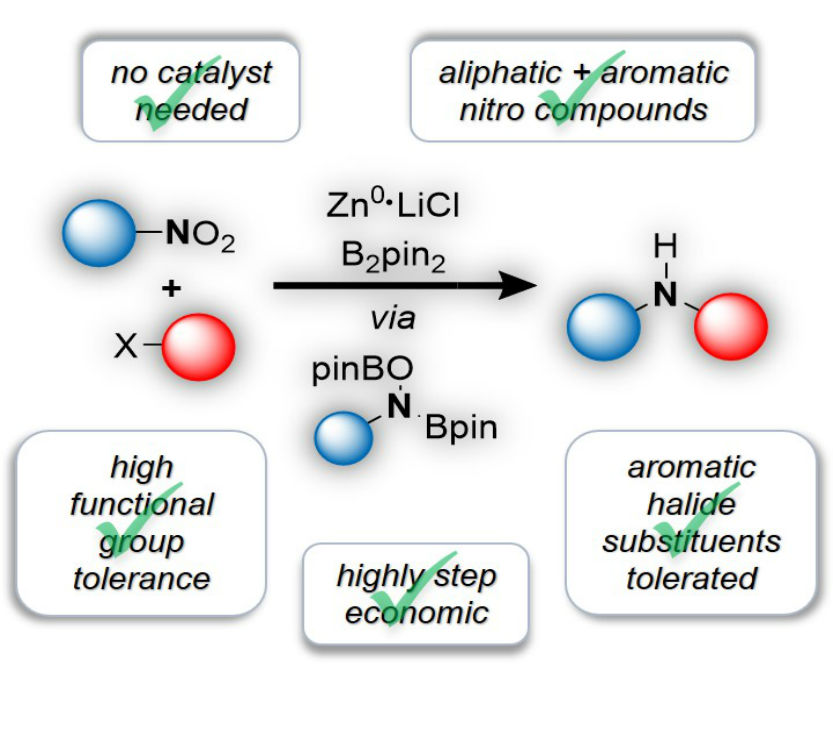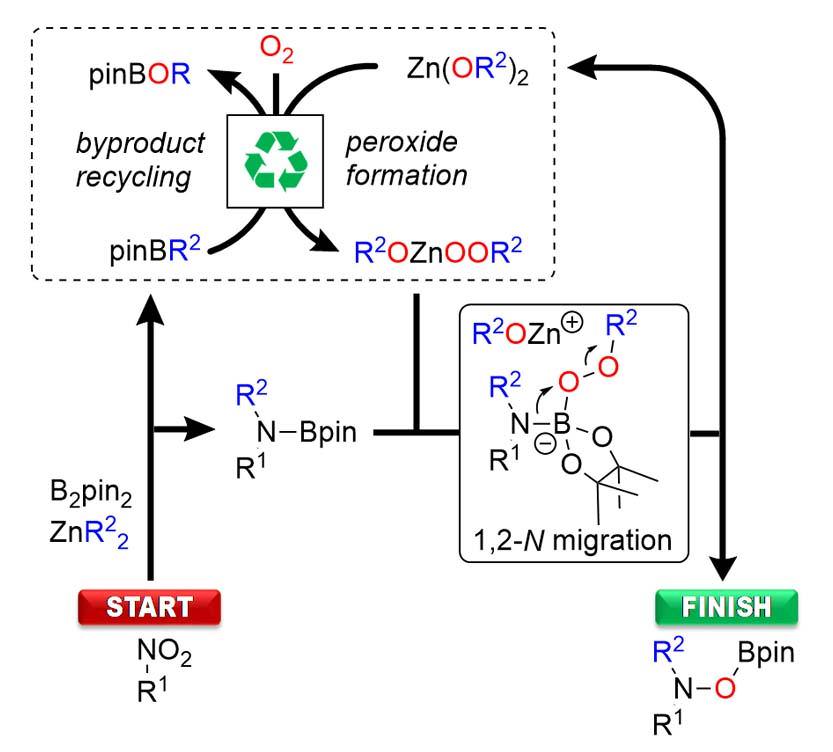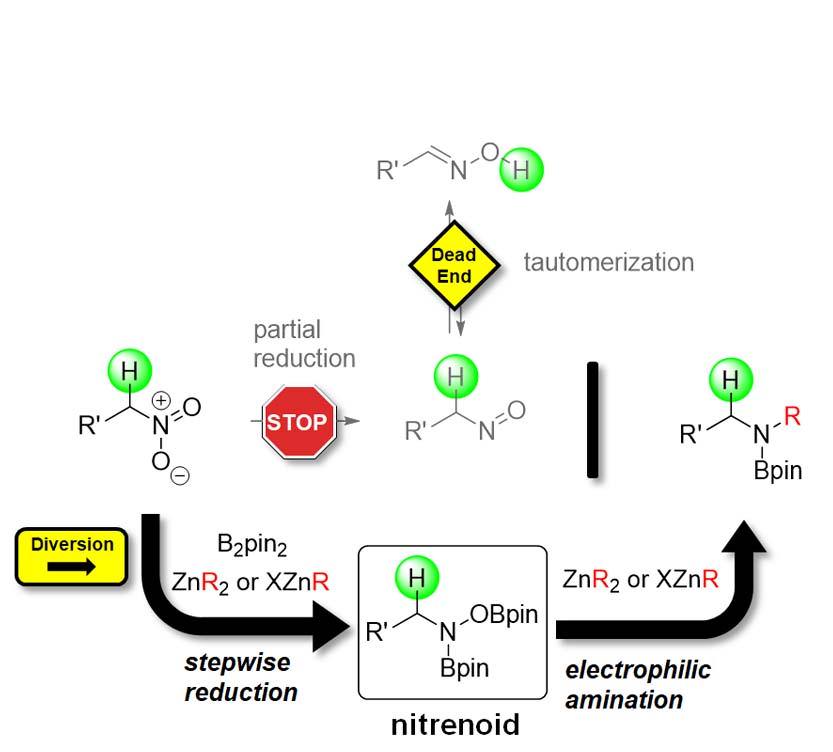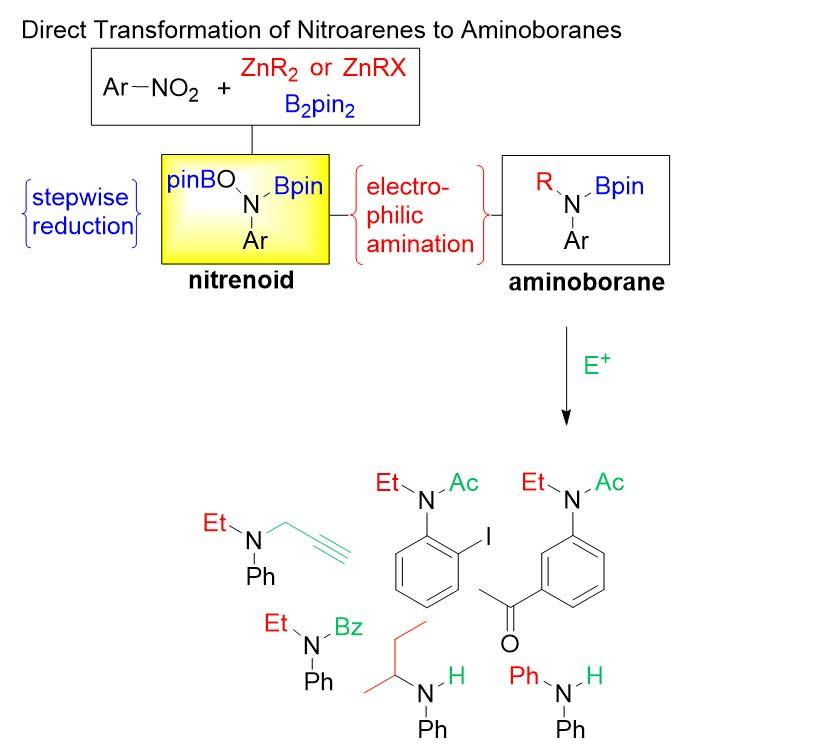
Boryl Anions
The stepwise reduction of nitro groups should provide access to compounds with various oxidation states at the nitrogen atom. These may then be used as reagents for the synthesis of functionalized amines. Seemingly straightforward, such a strategy has rarely been applied, as it comes with two major issues:
1.) Methods that halt the reduction at a defined oxidation state are underdeveloped.
2.) The intermediate oxidation states are often unstable and thus difficult to control.
As a solution to both, we have recently identified the prowess of boryl anions. Their reactivity is sufficient for nucleophilic addition to the relatively inert nitro group. At the same time the resulting borylated intermediates show significantly reduced sensitivity, making them stable enough to be engaged as electrophilic nitrogen reagents. Overall, this strategy represents a highly efficient shortcut, as commercially available nitro compounds are directly used as “umgepoled” nitrogen.